Julie Ahringer
Senior group leaderResearch summary
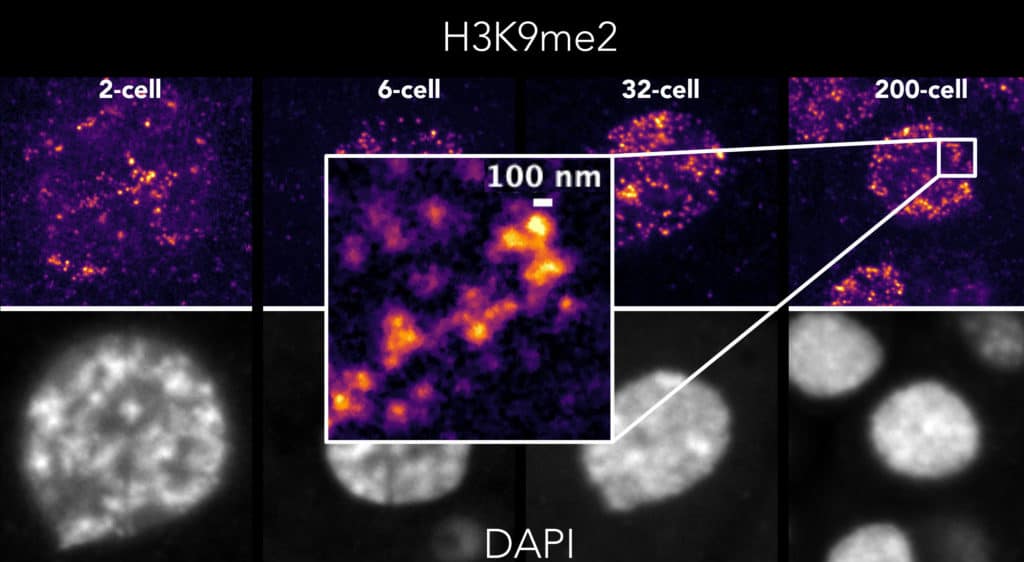
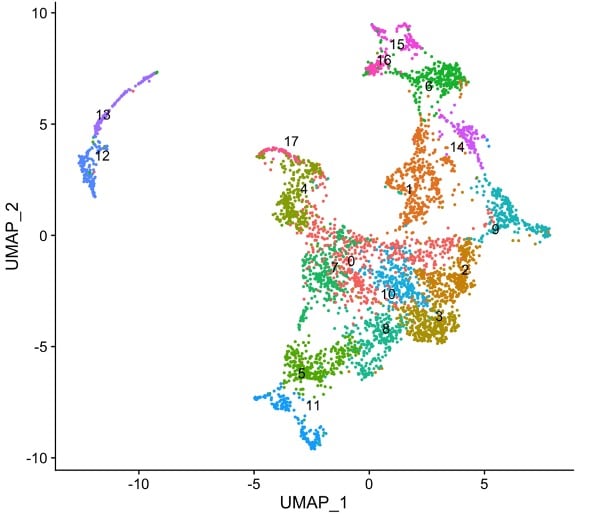
Developmental regulation of chromatin structure and function
Animal development is a remarkable process during which a single-celled totipotent zygote produces a myriad of different cell types. A driving force is the differential control of chromatin activity, which establishes gene expression programmes that drive cellular identity. Deciphering this control is necessary for understanding how the genome directs development and the diseases that result from chromatin dysregulation.

Selected publications
-
Gal C et al. (2021) DREAM represses distinct targets by cooperating with different THAP domain proteins. Cell Reports 37(3): 109835. DOI: 10.1016/j.celrep.2021.109835.
-
Serizay J et al. (2020) Distinctive regulatory architectures of germline-active and somatic genes in C. elegans. Genome Res 30: 1752-1765. DOI: 10.1101/gr.265934.120.
-
Jänes J et al. (2018) Chromatin accessibility dynamics across C. elegans development and ageing. eLife 7:e37344. DOI: 10.7554/eLife.37344.
-
McMurchy AN et al. (2017) A team of heterochromatin factors collaborates with small RNA pathways to combat repetitive elements and germline stress. eLife 6: e21666. DOI: 10.7554/eLife.21666.
-
Evans, KJ et al. (2016) Stable C. elegans chromatin domains separate broadly expressed and developmentally regulated genes. PNAS 113 (45): E7020–E7029. DOI: 10.1073/pnas.1608162113.
Biography
Professor Julie Ahringer PhD FRS FMedSci
Senior Group Leader, Wellcome Investigator, Professor of Genetics and Genomics, Member of the University Department of Genetics
Julie’s group investigates the regulation of chromatin and gene expression in development using molecular genetic and high-throughput genomic approaches.
Her lab pioneered genome-wide RNAi screening, carrying out the first systematic inactivation of genes in any animal by creating the widely-used C. elegans RNAi feeding library, and her earlier work focused on understanding embryonic cell polarity.
Julie got her first degree in Chemistry from Lafayette College in 1984, then did her PhD with Judith Kimble at University of Wisconsin-Madison. She came to Cambridge in 1991 as a postdoc with John White at the MRC-LMB, started her lab in the Genetics Department in Cambridge with a Wellcome Career Development Award in 1996, then moved in 1998 to the Wellcome CRUK Gurdon Institute (formerly called the Wellcome/CRC Institute), where she has been ever since.
She is a Fellow of the Royal Society and Academy of Medical Sciences, a member of EMBO, and received the Francis Crick lecture prize of the Royal Society.
Notable achievements and honours
-
2021Fellow of the Royal Society
-
2020George W. Beadle Award, Genetics Society of America
-
2007Fellow of the Academy of Medical Sciences
-
2006RNAi Innovator Award, Gene Expression Systems Ltd
-
2004Frances Crick Lecture Prize, Royal Society
-
2004Royal Institution Friday Evening Discourse
-
2003Member of European Molecular Biology Organisation
Research group
-
Dr Alex Appert
Research Associate
-
Szymon Berezicki
MPhil Student
-
Dr Toby Buttress
Research Associate
-
Dr Francesco Carelli
Research Associate
-
Yan Dong
Research Associate
-
Matthew Hill
PhD Student/ Research Assistant
-
Aleksander Klimczyk
MPhil Student
-
Dr Roopali Pradhan
Research Associate
-
Anna Townley
PhD Student
-
Dr Ser van der Burght
Research Associate