Martin Howard
Associate group leaderBuilding simple predictive models of complex biological processes
How are cellular memory states switched and maintained? Our group combines simple, predictive mathematical modelling with long- lasting experimental collaborations, to dissect biological mechanisms too complex to unravel by experiments alone. In many cases we are able to rationalise complex biological dynamics into simple underlying mechanisms, with few components and interactions. Our approach is highly interdisciplinary and relies heavily on the techniques of statistical physics and applied mathematics, as well as on close collaboration with experimental groups. This truly interdisciplinary approach allows us to get to the heart of biological mechanisms more speedily.

Selected publications
-
Lövkvist C et al. (2021) Hybrid protein assembly-histone modification mechanism for PRC2-based epigenetic switching and memory. eLife 10:e66454. DOI: 10.7554/eLife.66454.
-
Morgan C et al. (2021) Diffusion-mediated HEI10 coarsening can explain meiotic crossover positioning in Arabidopsis. Nat Commun 12(1):4674. DOI: 10.1038/s41467-021-24827-w.
-
Zhao Y et al. (2020) Temperature-dependent growth contributes to long- term cold sensing. Nature 583: 825-829. DOI: 10.1038/s41586-020-2485-4.
-
Yang H et al. (2017) Distinct phases of Polycomb silencing to hold epigenetic memory of cold in Arabidopsis. Science 357: 1142-1145. DOI: 10.1126/science.aan1121.
-
Berry S et al. (2017) Slow chromatin dynamics allow polycomb target genes to filter fluctuations in transcription factor activity. Cell Systems 4: 445-457.e8. DOI: 10.1016/j.cels.2017.02.013.
Biography
Professor Martin Howard DPhil, Associate Group Leader; Group Leader and Head of Department, Computational and Systems Biology, John Innes Centre
Martin Howard originally trained as a theoretical physicist, receiving his DPhil in 1996 from Oxford University. As a postdoc in Copenhagen, Vancouver and Leiden he then worked for several years on statistical physics. His interests then shifted towards biology and its interface with physics.
Following the award of a Royal Society University Research Fellowship, Martin moved to the Department of Mathematics, Imperial College London, where he stayed for nearly 5 years as a lecturer and then reader. In 2007, he moved to the Department of Computational and Systems Biology at the John Innes Centre as a professor and in 2020 was made an honorary group leader at the Babraham Institute, Cambridge.
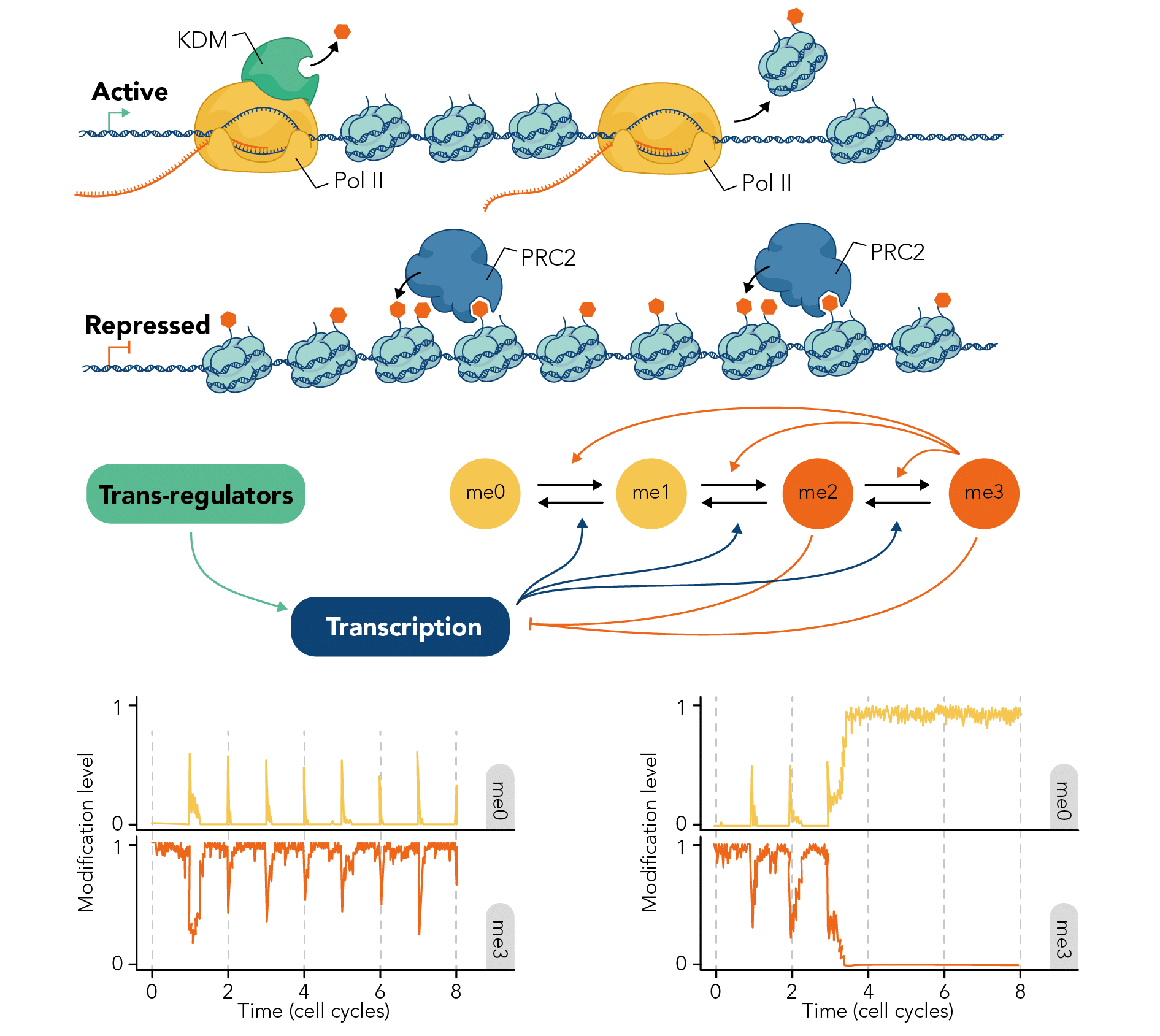
In the past he has worked on topics as diverse as cell size control, cell division positioning and metabolic resource allocation. A central focus of his current research is the mechanistic basis of epigenetic memory, fusing simple mathematical models with quantitative experimental data, where he has forged long-lasting collaborations with experimental colleagues. He has focused particularly on understanding Polycomb-based switching and memory, but also memory in the context of DNA methylation dynamics.
Notable achievements and honours
-
2020Institute of Physics Rosalind Franklin prize (shared with Physics of Life network committee)
-
2017-2023International Scientific Advisory Board of Max Planck Institute for Physics of Complex Systems (Dresden, Germany)
-
2014Institute of Physics Tom Duke Lecturer in Biological Physics
-
2008-2009Unilever Visiting Professor in Computational Science, University of Amsterdam
-
2002-2010Royal Society University Research Fellow