Ben Simons
DirectorResearch summary
Mechanisms of stem cell fate in tissue development, maintenance and disease
How do stem and progenitor cells regulate their fate behaviour to specify and maintain tissues? During development, cell proliferation and differentiation must be coordinated with collective cell movements to specify organs of the correct size, pattern and composition. In the adult, stem cells must regulate a precise balance between proliferation and differentiation to maintain tissue homeostasis.

Selected publications
-
Aztekin C et al. (2021) Secreted inhibitors drive the loss of regeneration competence in Xenopus limbs. Development 148 (11): dev199158. DOI: 10.1242/dev.199158.
-
Yum MK et al. (2021) Tracing oncogene-driven remodelling of the intestinal stem cell niche. Nature 594(7863):442-447. DOI: 10.1038/s41586-021-03605-0.
-
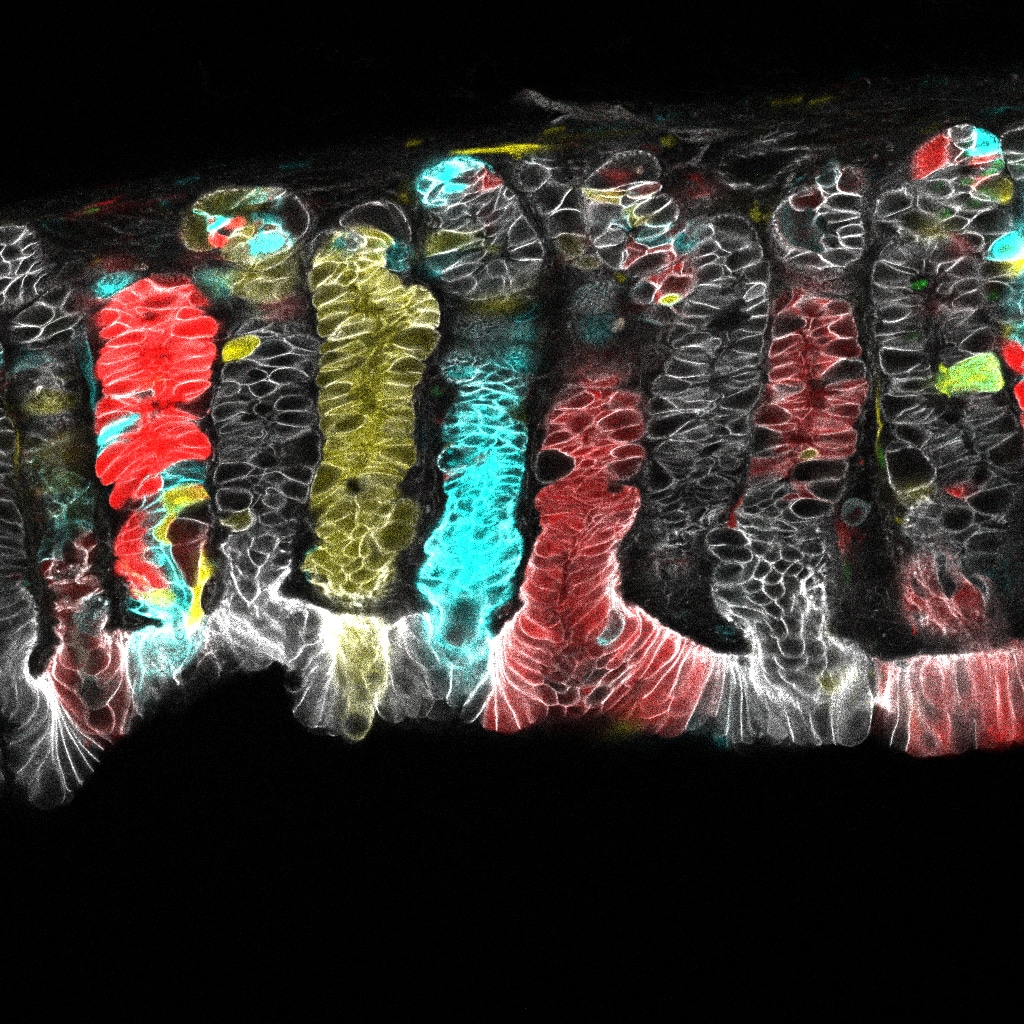
McGinn J et al. (2021) A biomechanical switch regulates the transition towards homeostasis in oesophageal epithelium. Nat Cell Biol 23(5):511-525. DOI: 10.1038/s41556-021-00679-w.
-
Nakamura Y et al (2021) Transient suppression of transplanted spermatogonial stem cell differentiation restores fertility in mice. Cell Stem Cell 28(8):1443-1456.e7. DOI: 10.1016/j.stem.2021.03.016.
-
Sznurkowska MK et al. (2020) Tracing the cellular basis of islet specification in mouse pancreas. Nat Commun 11(1):5037. DOI: 10.1038/s41467-020-18837-3.
-
Aragona M et al. (2020) Mechanisms of stretch-mediated skin expansion at single-cell resolution. Nature 584(7820):268-273. DOI: 10.1038/s41586-020-2555-7.
-
Hannezo E et al. (2017) A unifying theory of branching morphogenesis. Cell 171: 242–255. DOI: 10.1016/j.cell.2017.08.026.
Biography
Prof Ben Simons PhD FRS FMedSci
Director, Royal Society EP Abraham Professor, Herchel Smith Professor of Physics, Affiliate of the University Department of Applied Mathematics and Theoretical Physics
Research group
-
Dr Tal Agranov
Research Associate
-
Dr Lemonia Chatzeli
Research Associate
-
Joel Hochstetter
PhD Student
-
Dr Qiuyu Lian
Research Associate
-
Dr Ivan Lobaskin
Research Associate
-
Dr Sanne van Neerven
Research Associate
-
Dr Shay Melamed
Research Associate
-
Dr Laurien van de Weijer
Research Associate
-
Yanbo Yin
PhD Student