Unlocking the potential for clinical application of reprogrammed somatic cells by germinal vesicles from Xenopus meiotic oocytes
March 1, 2024
Read more
Correct body plan formation in Drosophila depends on the establishment of two axes of polarity in the developing oocyte. A new study by Doerflinger et al. shows that the antero-posterior axis requires the activation of Myosin II at the posterior of the oocyte, suggesting that cortical tension may act as the polarity cue.
Doerflinger H et al. (2021) The Drosophila anterior-posterior axis is polarized by asymmetric myosin activation. Current Biology DOI: 10.1016/j.cub.2021.11.024.
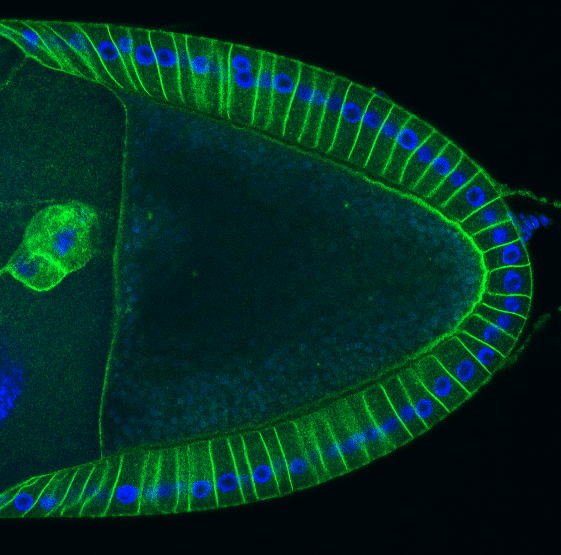
Par-1 protein trap endogenously tagged by GFP and DAPI
The Drosophila anterior-posterior (AP) axis is specified at mid-oogenesis when Par-1 kinase is recruited to the posterior cortex of the oocyte, where it polarises the microtubule cytoskeleton to define where the axis determinants, bicoid and oskar mRNAs localise. This polarity is established in response to an unknown signal from the follicle cells, but how this occurs is unclear.
Here we show that the myosin chaperone, Unc-45 and Non-Muscle Myosin II (MyoII) are required upstream of Par-1 in polarity establishment. Furthermore, the Myosin Regulatory Light Chain (MRLC) is di-phosphorylated at the oocyte posterior in response to the follicle cell signal, inducing longer pulses of myosin contractility at the posterior that may increase cortical tension. Over-expression of MRLC-T21A that cannot be diphosphorylated or treatment with the myosin light chain kinase inhibitor ML-7 abolish Par-1 localisation, indicating that posterior MRLC di-phosphorylation is essential for both polarity establishment and maintenance.
Thus, asymmetric myosin activation polarizes the anterior-posterior axis by recruiting and maintaining Par-1 at the posterior cortex. This raises an intriguing parallel with AP axis formation in C. elegans where MyoII also acts upstream of the PAR proteins to establish polarity, but to localize the anterior PAR proteins rather than Par-1.