Transposable elements recruited as promoters in the Caenorhabditis germline
December 16, 2022
Read more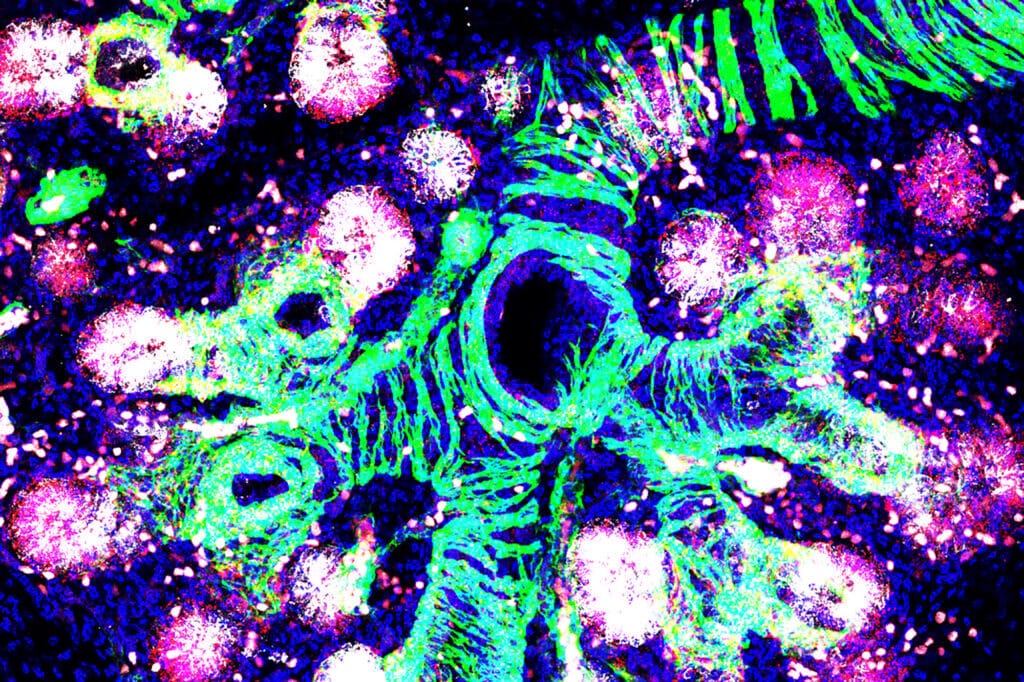
The study, led by the University of Leicester and University of Nottingham, and involving the Rawlins Lab (see image below), is the largest and most diverse study of its kind. The findings pave the way for potential new treatments to tackle conditions such as chronic obstructive pulmonary disease (COPD) and asthma, and highlight existing drugs that could potentially be repurposed at speed.
Published in Nature Genetics, the study analyses genome data from 580,869 participants worldwide to build the most confident picture yet of how our genes affect our lung health.
It is a huge boost to scientists as they seek to understand which medicines may help improve lung health, and also which medicines might make lung health worse.
Principal Investigator on the study, Professor Martin Tobin from the University of Leicester Department of Population Health Sciences, said: “This is a big leap in terms of the size and the ethnic diversity of the populations that we’ve been able to study before and it’s a huge step in the number of associated genetic variants that we’ve discovered.
Our genetic research findings can be used to generate individual risk scores that could personalise medicine. At this stage the risk scores we developed form important tools for further research, but in the future these could help to select which drugs might be most effective for individual patients, and which drugs should be avoided.”
Shrine, N., Izquierdo, A.G., Chen, J. et al. Multi-ancestry genome-wide association analyses improve resolution of genes and pathways influencing lung function and chronic obstructive pulmonary disease risk. Nat Genet 55, 410–422 (2023). DOI: 10.1038/s41588-023-01314-0

The image on the cover was provided by co-author Emma Rawlins and taken by Gurdon Research Associate Kyungtae Lim. It shows a 16 weeks post-conception human fetal lung. The branching airway tree is surrounded by bands of developing smooth muscle (green), which also labels the developing vasculature. The epithelial progenitor cells (co expressing SFTPC in white and NOTUM in red) are initiating the process of alveolar epithelial development.