How polarised epithelial cells grow “up” without breaking the barrier
September 28, 2022
Read more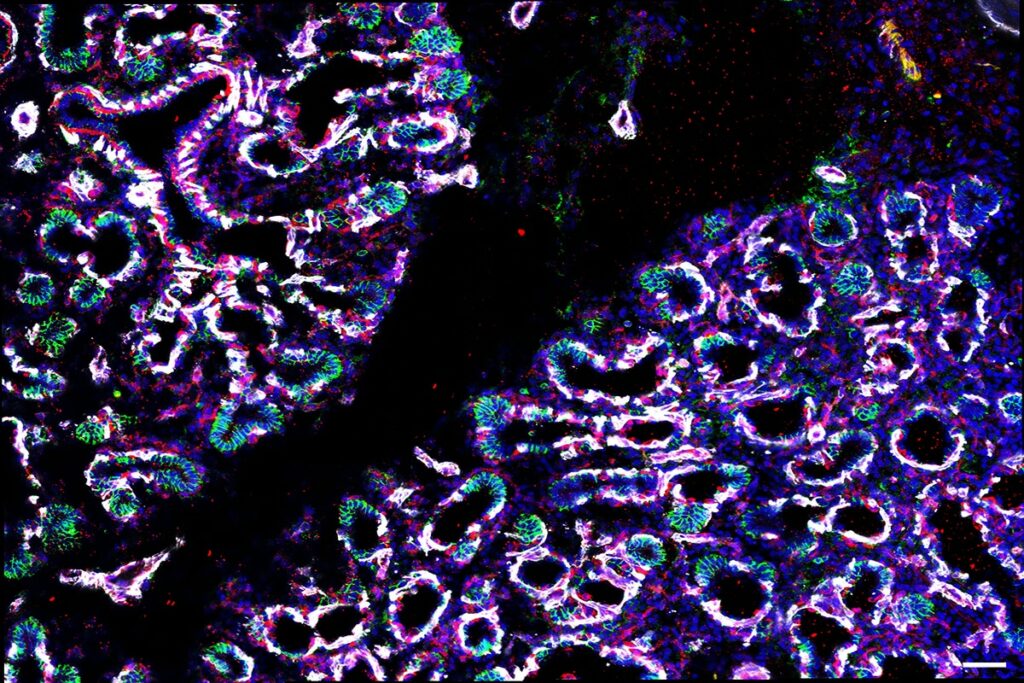
A study from the Rawlins lab led by Kyungtae Lim has deciphered the role of cell-cell interactions in building the human lung gas exchange surface. The lab developed a new organoid system which also allowed them to identify transcription factor targets and to perform disease modelling. This has been a highly collaborative project, including with Alex Donovan from the Brand lab.
Lim K., Donovan A.P.A., Tang W., Sun D., He P., Teichmann S.A., Marioni J.C., Meyer K.B., Brand A.H., and Rawlins E.L. (2023). Organoid modelling of human fetal lung alveolar development reveals mechanisms of cell fate patterning and neonatal respiratory disease. Cell Stem Cell 30, 1-18. Published online December 8, 2022. DOI: 10.1016/j.stem.2022.11.013.
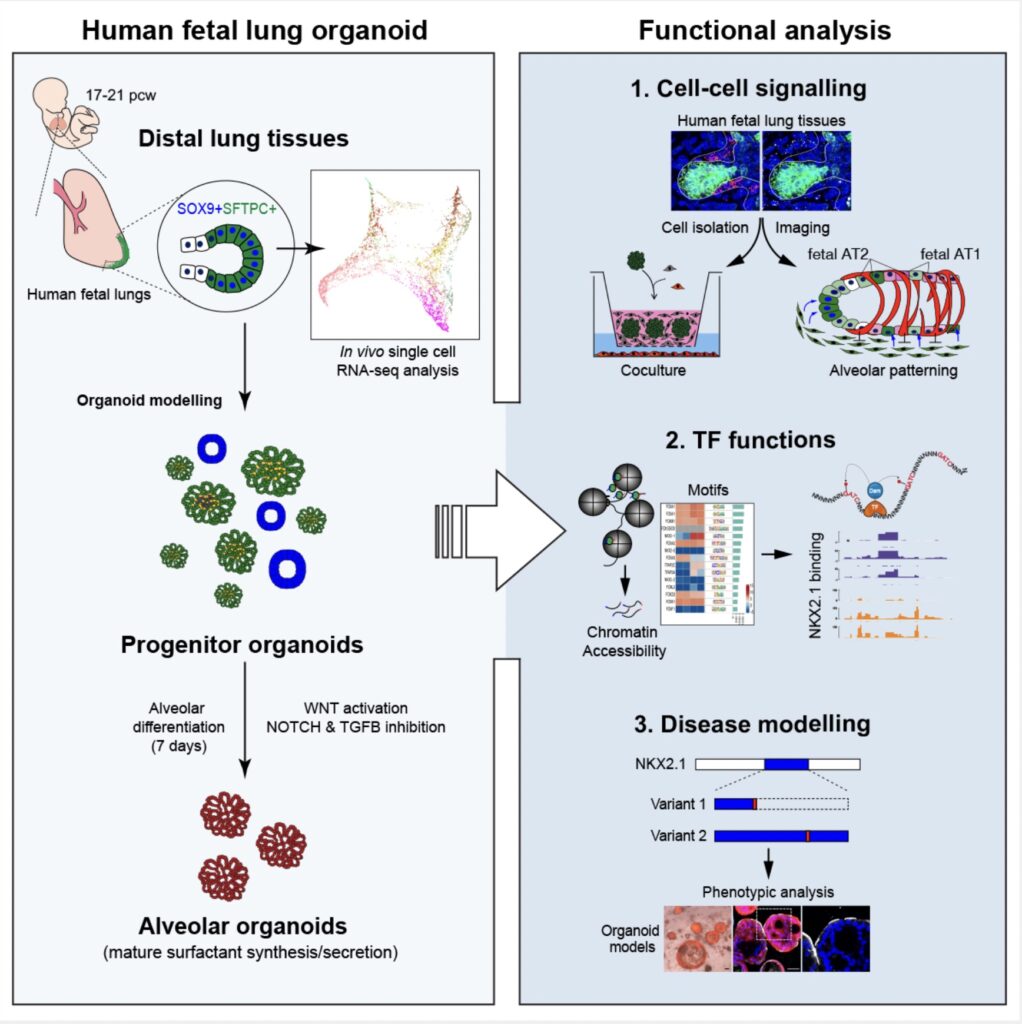
Variation in lung alveolar development is strongly linked to disease susceptibility. However, underlying cellular and molecular mechanisms are difficult to study in humans. We have identified an alveolar-fated epithelial progenitor in human fetal lungs, which we grow as self-organizing organoids that model key aspects of cell lineage commitment. Using this system, we have functionally validated cell-cell interactions in the developing human alveolar niche, showing that Wnt signaling from differentiating fibroblasts promotes alveolar-type-2 cell identity, whereas myofibroblasts secrete the Wnt inhibitor, NOTUM, providing spatial patterning. We identify a Wnt-NKX2.1 axis controlling alveolar differentiation. Moreover, we show that differential binding of NKX2.1 coordinates alveolar maturation, allowing us to model the effects of human genetic variation in NKX2.1 on alveolar differentiation. Our organoid system recapitulates key aspects of human fetal lung stem cell biology allowing mechanistic experiments to determine the cellular and molecular regulation of human development and disease.